Unfortunately, humans and mice are both mammals and many of our proteins/receptors share high homology. Finally, nearly all the current therapeutic mAbs are of murine origin, largely thanks to the hybridoma technology that enables the mouse B-cells to be immortalized and screened in clonal fashion for antigen-specific antibodies. Antibodies are also difficult to be manipulated for drug conjugation via the conventional conjugation and linker chemistries, as they are too big to be synthesized chemically and too complex to be produced in microorganisms. The astronomically high cost associated with therapeutic antibody production makes mAb-based therapeutics out-of-reach to most of the world’s population. In addition, most mAbs have to be produced in mammalian cells and often require post-translational modifications, such as specific glycosylation patterns. Despite its important role in prolonging the antibody half-life and recruiting immune effector cells, the antibody constant region-Fc-can sometimes give rise to adverse effects, such as antibody-dependent-enhancement (ADE) of infection by some viruses e.g., Dengue virus and Zika virus (Screaton et al., 2015 Dejnirattisai et al., 2016 Paul et al., 2016). For example, the large size of antibodies (~150 kDa) may impede their ability to penetrate into tumor tissue (Chauhan et al., 2011 Shah and Betts, 2013), and the planar binding interface makes it difficult to obtain antibodies that bind to grooves and catalytic sites of enzymes (Skerra, 2000). However, antibodies are not without their limitations. Since then, over 62 mAbs have been approved by the FDA as therapeutics and many new candidates are presently undergoing preclinical and clinical evaluations (Ecker et al., 2015 Reichert, 2017). The first monoclonal antibody (mAb) entered human therapy in 1986. Additional advantages and limitations of these protein scaffolds as therapeutics compared to antibodies will be discussed.Īntibodies have long been regarded as ‘magic bullets’ in human therapy due to their ability to bind targets with high affinity and specificity (Strebhardt and Ullrich, 2008). In addition, many of these scaffolds derive from human proteins and thus possess very low immunogenic potential. Most of these scaffolds are thermostable and can be easily produced in microorganisms or completely synthesized chemically. Scaffolds that fall under the first category include adnectins, anticalins, avimers, Fynomers, Kunitz domains, and knottins, while those belonging to the second category include affibodies, β-hairpin mimetics, and designed ankyrin repeat proteins (DARPins). Based on the location of the amino acids that mediate ligand binding, we place all the protein scaffolds under clinical development into two general categories: scaffolds with ligand-binding residues located in exposed flexible loops, and those with the binding residues located in protein secondary structures, such as α-helices. Although not all are expected to be approved, the significant benefits ensure that these molecules will continue to be investigated and developed as therapeutic alternatives to antibodies. 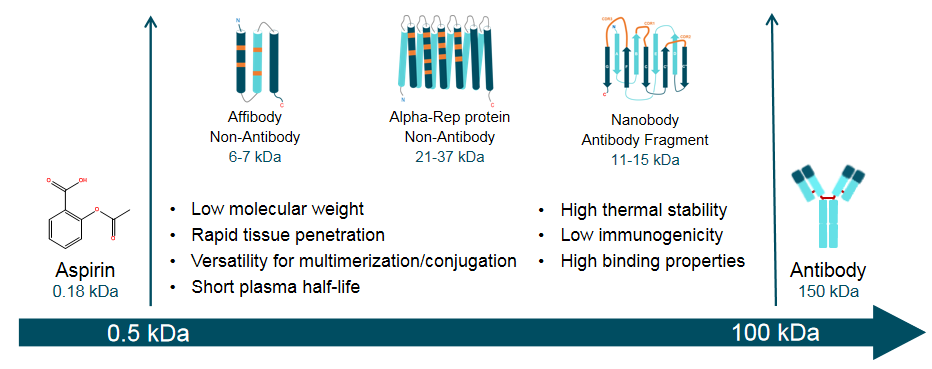
This review focuses on protein scaffolds that are either already being used in humans or are currently being evaluated in clinical trials. New binding protein scaffolds are constantly being designed or discovered with some already approved for human use by the FDA. Therefore, non-antibody binding proteins have long been sought after as alternative therapies. Unfortunately, antibodies are also limited in several respects, chief amongst those being the extremely high cost of manufacture. Antibodies have proved to be a valuable mode of therapy for numerous diseases, mainly owing to their high target binding affinity and specificity. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
